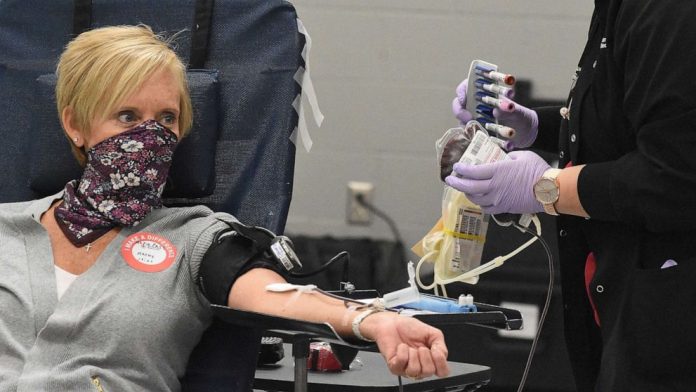
Starting next week, the American Red Cross will offer antibody tests for people who suspect they were previously infected with the novel coronavirus and are interested in donating their blood plasma — a potential game-changer in the treatment for seriously ill COVID-19 patients.
“That completely changes the landscape,” Dr. Pampee Young, chief medical officer for the American Red Cross, told ABC News in an interview Wednesday.
Antibody tests can indicate whether a person has been exposed to the novel coronavirus and has recovered, developing the antibodies to fight it. And while there is no guarantee that antibodies to this new virus actually provide immunity, doctors are hopeful that patients severely sickened with COVID-19 can benefit from infusions of blood plasma collected from those who have recovered from the disease. The therapy, known as convalescent plasma, is a century-old technique used for treating epidemics.
At the moment, an individual who wishes to donate blood plasma for the experimental convalescent plasma therapy must have documentation of a positive COVID-19 test. The lack of diagnostic tests available has led hospitals and donation centers to say they are in desperate need of donors.
“Qualifying and getting the right donors into our centers to donate is one of the biggest hurdles in this in this endeavor,” Young said.
More than 30,000 people have requested to donate on the American Red Cross website, but only 2 to 3% actually qualify and meet the current criteria set by the U.S. Food and Drug Administration. That donor pool could increase dramatically with the implementation of antibody testing at American Red Cross donation centers.
“All of those (potential) donors who don’t have confirmed testing can now be tested,” Young said. “We can really cut through that time, which is so important.”
Young explained that researchers and medical professionals have been in uncharted territory, “building the plane as you’re flying it.” But she said they’ve been working around the clock to streamline the process.